
Chemi-Pharm AS develops, produces and sells allergen-free disinfection agents, cleansing and skincare products and detergents. Products are exported to more than 25 countries worldwide; the company’s main customers are in medical sector.
Chemi-Pharm product portfolio also includes
skincare line D’DIFFERENCE and
health and pharmaceutical brand Medihex.
PRODUCT CATEGORIES
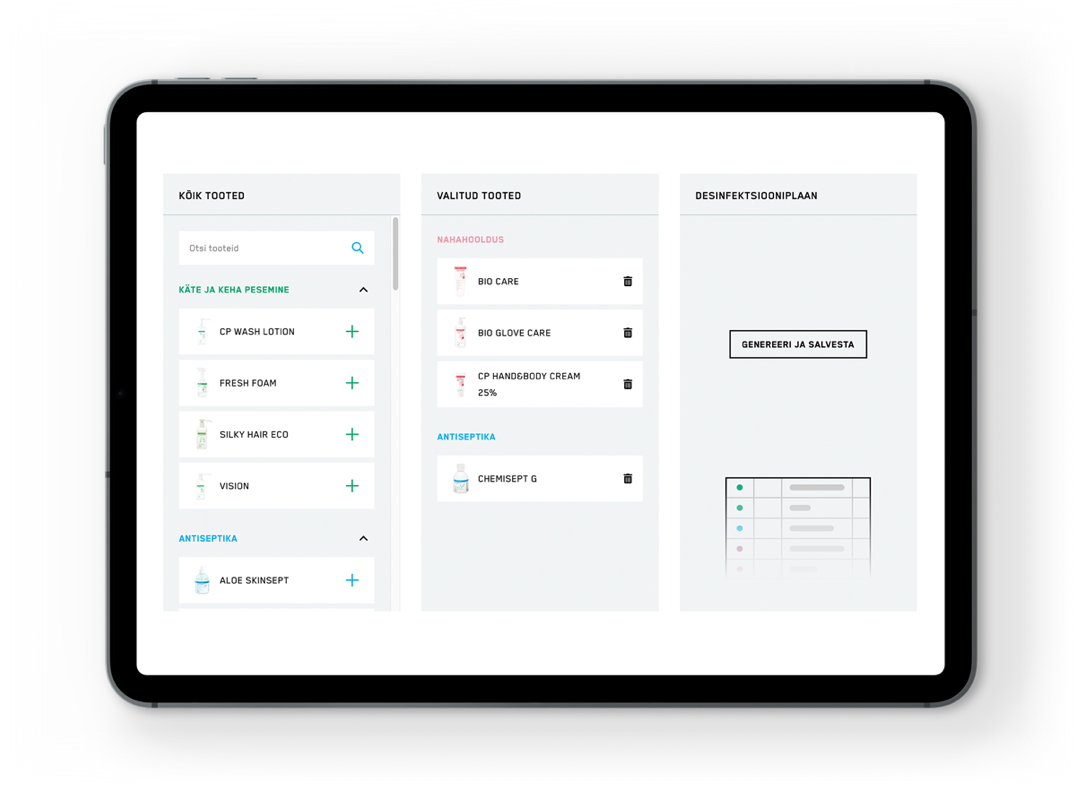
Cleaning and disinfection plan
Create a cleaning and disinfection plan with Chemi-Pharm products.
OUR BRANDS

D’DIFFERENCE is the science of skincare. Invented by an MD, developed by chemists in our own labs, and produced with cold-mixing technology, our active ingredients are 100-percent natural.
D’DIFFERENCE is a natural version of products originally created for use by physicians to solve acute clinical problems. True to our medical roots, D’DIFFERENCE products are formulated through evidence-based results from extensive testing, yet our Nordic heritage means we embrace the natural, living as part of nature, never separate.
In this age of eco, it seems everyone with a kitchen is creating skincare products. But we take pride in the fact that ours are backed by science, meet ISO 9001 production standards, and leave the skin healthy both inside and out.
Medihex is a science-based health and pharmaceutical brand with a focus on creating a sense of security and well-being for people. Medihex’s products are high-tech, born of collaboration between researchers, universities and recognized companies.
News
See all the news2023 STABILITY IS SHOWING SIGNS OF RETURN
THE BIOBLOCK® NASAL SPRAY CLINICAL STUDY
CHANGES IN CHEMI-PHARM AS MANAGEMENT STRUCTURE
THE NORDIC SWAN ECOLABEL An eco-certificate recognized in the Nordic countries!
CHEMI-PHARM IS PARTICIPATING IN A DIGITALIZATION SUPPORT PROGRAM BY ENTERPRISE ESTONIA
ESTONIAN HEALTH TECHNOLOGY COMPANIES ATTENDED A BUSINESS MISSION IN SAUDI ARABIA
GREEN ICT

With the help of the estonian-norwegian cooperation project green ict, chemi-pharm’s production process will be more efficient and there will be a remarkable increase of units produced within the same working hours.